- Next to T. B. Hospital Main Gate, Koch’s House, Jerbai Wadia Road, Sewri
Radionuclide Therapy
- These are FDA-approved have been in routine clinical practice.
- Commonly performed radionuclide therapies are:
– Radioiodine therapies for thyroid cancer
– Radioligand therapy for metastatic prostate cancer
– PRRT for metastatic neuroendocrine tumors
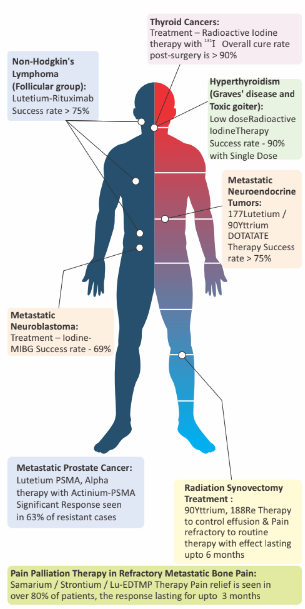
How effective are radionuclide therapies?
Radionuclide Therapy
- RADIOIODINE THERAPY IN THYROID CANCER
- LUTETIUM PSMA IN METASTATIC PROSTATE CANCER
- LUTETIUM PRRT IN METASTATIC NET
- METASTATIC NEUROBLASTOMA
- BONE PAIN PALLIATION
Success Rate
- >90% CASES POST-SURGERY
- 63% EVEN IN CASES RESISTANT TO CONVENTIONAL THERAPIES
- >75%
- 69%
- 80% (with single dose response lasting for upto 3 months)
RADIOIODINE THERAPY FOR THYROID CANCER
Indications
- Remnant ablation in a post-thyroidectomy setting
- Treatment of metastatic thyroid cancer
- Treatment of recurrent disease in thyroid bed on follow-up
- Radioiodine is a colourless, tasteless water-like liquid that is administered orally
No major side effects of the procedure.
PEPTIDE RECEPTOR RADIONUCLIDE THERAPY
Who would benefit from PRRT ?
- Adults with metastatic / inoperable neuroendocrine tumor
How effective is PRRT in metastatic NET ?
- As per published literature, PRRT causes a 79% reduction in risk of disease progression.
- PRRT reduces the tumour growth and achieves a stable growth-free period ranging from 1-4 years even after finishing the therapy
- It has already been used in over 8000 patients, predominantly in the Europe
- PRRT is an FDA approved therapy in the USA.
What are the side effects ?
- Minor Only.
RADIOLIGAND THERAPY FOR METASTATIC PROSTATE CANCER
- Therapy for metastatic prostate cancer has shown very promising results.
- There are no major side effects to the treatment.
RADIONUCLIDE THERAPY FOR BONE PAIN PALLIATION
- Beta emitting radioisotopes like Lutetium / Samarium selectively binds to these bone lesions in higher concentration and cause pain relief.
Single intravenous injection shows an objective response in over 80% of these patients.
Targeted CANCER Treatment with NUCLEAR Medicine Therapy
Radionuclide Therapies
- Radionuclide therapies involve the administration of radioactive isotopes to the patient that binds to the target cells and emits radiation selectively to those specific tumor cells.
- Therapies like Radioiodine for thyroid cancer and the more recent PRRT with Lutetium DOTATATE are FDA-approved and have been in routine clinical practice.
- Commonly performed radionuclide therapies are:
- Radioiodine therapies for thyroid cancer
- PRRT for metastatic neuroendocrine tumors
- Radioligand therapy for metastatic prostate cancer